Our diet is critical for our health. In the past several years, interest has increased in food additives and dietary supplements such as prebiotics like β-galactooligosaccharides (GOSs). The determination of total GOS contents in food and supplements is essential to fulfill strict food labeling and safety requirements. The most widely used method for total GOS determination is based on enzymatic hydrolysis to break down the complex molecules into simple carbohydrates prior to their chromatographic analysis. This article outlines the advantage of using an improvement to AOAC Method 2001.02 using ion chromatography with amperometric detection (IC-PAD) and full sample automation after enzymatic hydrolysis.

What are GOSs?
GOSs are chains of galactose units with an optional glucose end. They are often naturally present in small amounts in various foods and beverages.
Initially discovered as major constituents of human breast milk (present up to 12 g/L), GOSs are added as a prebiotic supplement to infant formulas. They show bifidogenic effects, meaning they support growth and well-being of non-pathogenic gut bacteria.
GOS supplements are available either raw, or as concentrated powders or syrups, and are subsequently used by food manufacturers to enrich consumer products or sold as supplements.
GOS labeling requirements
The ongoing growth of global prebiotic and GOS markets is a result of increasing consumer awareness regarding healthy eating. Similarly, increased demand regarding food quality has led to stricter, more comprehensive rules for food labeling and safety (e.g., EU 1169/2011 and EU 2015/2283). The determination of total GOS contents in food, supplements, or raw products is thus essential to fulfill such requirements.
Studies about GOS health effects recommend maximum doses under 30 g per day, though this is much stricter for infant formulas. Otherwise, there are no other limits regarding GOS content in food or as nutritional supplements.
AOAC 2001.02
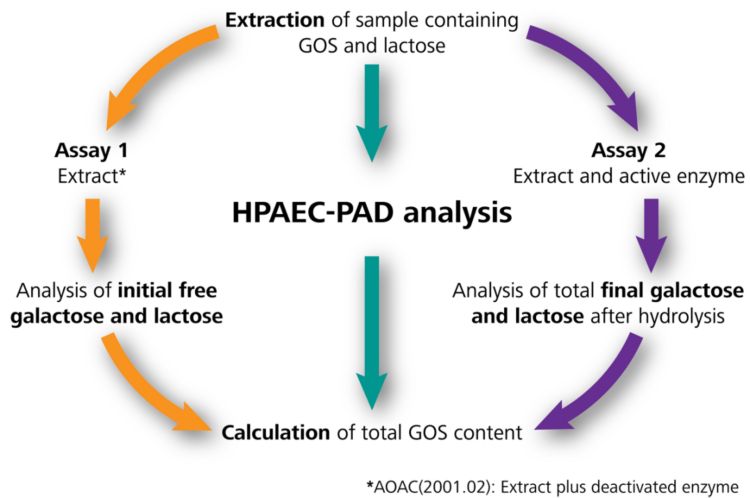
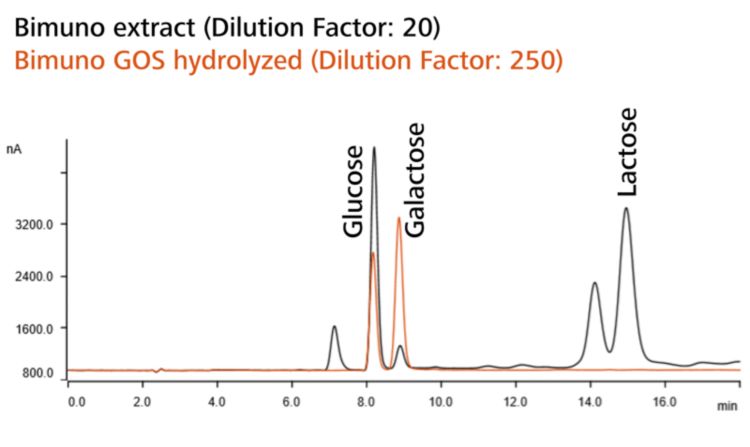
The most widely used method to measure total GOSs in food products is the standard method AOAC 2001.02. This method is based on the extraction of GOS from a sample followed by enzymatic hydrolysis of the oligosaccharides into monosaccharides and their subsequent analyses with high performance anion exchange chromatography with pulsed amperometric detection.
In AOAC, chromatography for anions is referred to as HPAEC (high performance anion exchange chromatography) but here we will simplify this to the generic term of IC.
The key to AOAC 2001.02 is the comparison of a control solution with one which has been treated and hydrolyzed with an enzyme (β-galactosidase). The enzyme catalyzes the splitting of glycosidic bonds and hydrolyzes GOSs and lactose into glucose and galactose. The concentration differences of free galactose and lactose determined in these two solutions is used to calculate the total GOSs (Figure 1).
Improvements to the AOAC Method
The sample preparation for AOAC 2001.02 is rather complex: one shortcoming is the incubation of the reference solution with the deactivated enzyme (which is rather expensive) to determine the initial carbohydrate concentrations (Figure 1) rather than using the pure extract. Another critical point is the sample dilution procedure, which is supposed to be done in acetonitrile, while standards are based on ultrapure water.
Here, the focus was to simplify the entire procedure to increase the ease of use and the overall efficiency of the method.
The improved method for total GOS content analysis uses the extract for measuring of the initial glucose, galactose, and lactose concentrations (Figure 1 Assay 1). However, the deactivated enzyme was not used, and instead comparisons were made to see if its presence had any effect on the results. This step was eliminated after proving results equivalent to AOAC 2001.02 Assay 1 (with the deactivated enzyme), but chemical expenses and additional manual work are reduced. The total GOS content is therefore calculated from the analyte concentrations in Assay 1 (without any enzyme) and Assay 2 (extract with the active enzyme) (Figure 2).
Want to know more details about the application? Download our free Application Note below about total GOS analysis in foods with ion chromatography.
Quality labels for novel foods – Improvement on AOAC 2001.02: GOS analysis with IC-PAD
Aside from the enzyme usage, the official AOAC method for analysis of total GOSs suggests that standards be prepared in ultrapure water (UPW) while samples are to be diluted with 20% acetonitrile. A control experiment was performed to compare results between:
- Dilutions in UPW evaluated with UPW calibration (“UPW option”)
- Dilutions in acetonitrile evaluated with UPW calibration (AOAC 2001.02)
- Dilutions in acetonitrile evaluated with acetonitrile calibration (“ACN option”)
Reproducibility of total GOS contents was compared among the three options, with the UPW and AOAC preparation options exhibiting similar results. The ACN option resulted in lower total GOS contents than the others. Additionally, the acetonitrile did not seem to lend a stabilizing effect to the samples. This supports the improvement of the AOAC method by performing sample dilutions with UPW instead of acetonitrile, saving unnecessary reagents and limiting the chemical imprint of the analysis.
Results
Overall, the satisfying variability, target and spike recoveries, together with the interference tests proved the modified method as valuable and robust. With limits of detection (LODs) of 0.1 mg/L (galactose) and 0.2 mg/L (glucose, lactose) in solution, even low total GOS contents can be determined with high precision.
Summary
As a multicomponent method, ion chromatography with amperometric detection is a very selective, sensitive, and robust analysis method for carbohydrates without any additional derivatization steps. In combination with enzymatic treatment, even more complex carbohydrates can be quantified.

This research presents an update to the standard AOAC method for total GOS determination in foodstuffs. With the same principle (enzymatic hydrolysis of complex GOS molecules followed by chromatographic analysis of simple carbohydrates), analytical method efficiency was improved in favor of laboratory time and running costs. Additional automation steps (e.g., Metrohm Inline Dilution and automatic calibrations) can further improve the method efficiency.
Want more information about the simplified method for total GOSs via IC-PAD? More details about the improvement of AOAC method 2001.02 by reducing manual laboratory work and eliminating expensive reagents can be found in our article published in The Column from LC/GC (2021): Improving on AOAC 2001.02: GOS Determination in Foods Using HPAEC–PAD.
 Share via email
Share via email
