Why consider automation – even for simple titrations
Apr 27, 2020
Article
If you are reading this blog post, you are most likely already familiar with the general principles of potentiometric titration. Although chromatographic and spectroscopic methods are preferred in many labs, titration is still the method for analysis of all kinds of sample types. Titration stands apart from other techniques because it is an absolute method (also known as a primary method). Whenever the analyte reacts in a known stoichiometric way with another reagent, titration is the method of choice, not only for official norms and standards.
Nowadays, titration is far more modern than it was back when I was a student. At that time we still used glass burets and color indicators, and suffered from inconsistent results. Although the automatic addition of the titrant and the recognition of the equivalence point are now performed by the titrator itself in most labs, there are still many manual steps that can go wrong and lead to unreliable results.
How to avoid titration errors in your lab
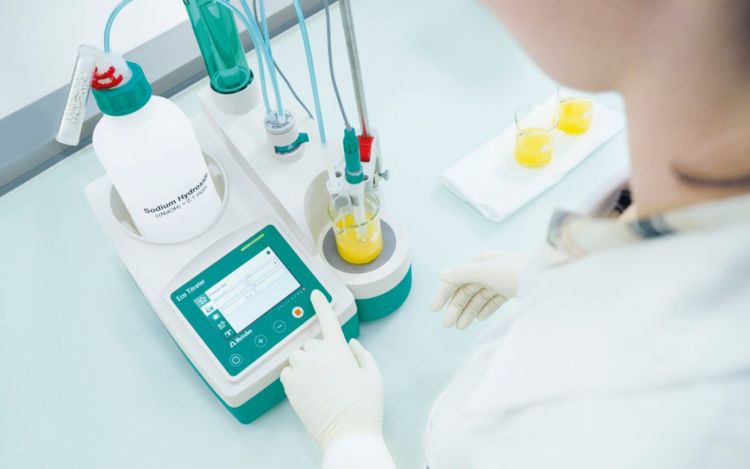
If the used titrator is a stand-alone type, the analysis becomes a full-time job for the lab technician. Not only must the sample be prepared, the titration itself has to be started after the sensor and buret tip have been placed in the sample solution. If using a titrator, the addition of the titrant as well as evaluation and calculation of the results will be done automatically. However, cleaning after each finished determination and preparation for the next sample still remains the task of the lab technician. In many cases, the titration does not take much longer than 3–5 minutes. Due to this short period, there are not many other tasks which can be completed by the technician during the analysis time.
Using a fully automated titration system results in not only more efficient analyses, but achieves better and even more reproducible ones at the same time.

Save valuable time
Time savings is one of the biggest benefits of using automation. To get a better idea about the general amount of time that can be saved, have a closer look at this diagram. You can already see how many steps can be done by an automated titration system, leaving analysts more time for other tasks.
A proper analysis starts with the correct liquid handling

Sample determination in titration can consist of several manual steps beyond the addition of the titrant. Depending on the type of sample and analysis, different kinds of sample preparation steps are required. The most common ones are the manner of sampling itself, dilution, auxiliary reagent addition, pH, or temperature adjustment.
Taking the correct amount of representative sample can already be quite a demanding task. For many applications, the sample is weighed if it is solid (e.g., powder or tablets), but this does not work for all sample types. Liquids are normally measured using measuring cylinders or pipets. These are very accurate and helpful tools if the user knows how to handle them correctly.
As long as the same person is performing the sampling, the results should be very reproducible, but in most labs this is not the case. Usually more than one person is responsible for the same analysis due to shift work, which can result in differing or less reproducible results.
With fully automated volumetric sampling, the only thing you need to care about is making sure enough sample is placed in the sample beaker! The connected dosing device is able to pipet the requested sample amount very accurately to the titration cell. The big advantages of using an automated pipetting system is its flexibility. Due to its independence of the user, the sampling and the results become much more reproducible.

Dilution / Addition of auxiliary reagents
In many cases, the sample amount needed for the analysis is not sufficient enough to put the sensor directly in and begin analysis. Often deionized water (or another suitable solvent) is added to reach a sufficient volume for the sensors to be placed in. As titration is an absolute method, the amount of added solvent has no impact on the titration results, as long as the solvent does not react in the same way as the sample does with the titrant.
A typical example is the solvents used for TAN/ TBN analysis in the petrochemical industry. Here, it is important to measure the amount of added solvent accurately and make sure to determine the blank value in advance.
Determination of the total acid number in petroleum products
Determination of the total base number in petroleum products
There are quite a few other applications where an accurate amount of reagent must be added: e.g., to start or stop a reaction, preparing back titrations, or for general pH adjustments before the final titration can take place.

For these tasks, measuring cylinders and pipets are normally used, but this is often tedious and can lead to mistakes, especially if many samples have to be analyzed. These days, many stand-alone titrators already offer the possibility to automatically and accurately add reagents, including the titrant. Repetitive (and annoying!) manual preparation steps therefore no longer occupy the lab technician.
Since reagent addition is part of the sample determination procedure, these added volumes can be documented much easier and more accurately, meaning less trouble when it comes to the analysis procedure traceability.
So, how good can such a buret be? Metrohm offers burets with a resolution of 100,000 pulses where even minute volumes can be dosed with extreme accuracy. For example, when using a 50 mL cylinder unit we are speaking about 0.5 µL per pulse.
The best liquid handling is not good enough if the sensor measures incorrectly
The heart of each titration or measurement is the chosen sensor. It is especially important in potentiometric titrations that both measuring and reference electrodes are properly cleaned, and if necessary, also conditioned between analyses. Otherwise, false equivalence points might be indicated, or instable curves will be shown, which leads to inaccurate and unreproducible results. Therefore, proper sensor maintenance is also important. Although many lab technicians are trained about handling the electrode correctly, some things may be forgotten after some time and this is where the trouble starts.
Quite often it takes some time before realizing that the wrong electrode treatment is the reason for the differing results. Several issues could be cleared up due to either the absence of electrode cleaning/conditioning or perhaps the cleaning step was not long enough. Similar to the titration itself, the manual cleaning steps also depend on the user performing this task. With an automated setup, this can be easily avoided as the electrode is treated in the exact same way for each determination. Additionally, automating the titration guarantees that the sensor is always properly stored, even if the sample series finishes in the middle of the night when no one is in the lab to do this.
In the blog entry below, you will find more useful hints for correct sensor handling in general.
Avoiding the most common mistakes in pH measurement
Last but not least, a well-treated electrode not only gives you outstanding results, but also lasts much longer and reduces the costs of consumables.
Automation rocks – even for simple titration applications
Here I have explained several reasons to consider automation even for simple titration applications. By including as many sample preparation steps as possible directly into the analysis run, this guarantees that each sample is treated exactly the same way, along with a better documentation process. Not only is there a reduction in handling errors during sampling, liquid handling, and electrode treatment, but as a result of these the reproducibility will be increased. On top of this, lab technicians are no longer occupied with annoying routine sample preparation/determinations, but have more time for reporting tasks or other analyses which are not automated – i.e., the laboratory throughput increases.
 Share via email
Share via email