
AN-V-222
2020-05
Iron determination in drinking water
Wide linear range with a low detection limit using the Bi drop electrode and the triethanolamine method
Summary
The presence of iron in drinking water can lead to an unpleasant, harsh metallic taste or reddish-brown stains. In addition, «iron bacteria» which can grow in waters containing Fe as low as 0.1 mg/L, create a reddish-brown slime that can clog plumbing and cause an offensive odor. Over a longer period, the formation of insoluble iron deposits is problematic in many industrial and agricultural applications, such as water supply, system cooling, or field irrigation. To avoid these problems, the U.S. Environmental Protection Agency (EPA) defines the Secondary Maximum Contaminant Level (SMCL) for water treatment and processing plants as 0.3 mg/L Fe in drinking water.
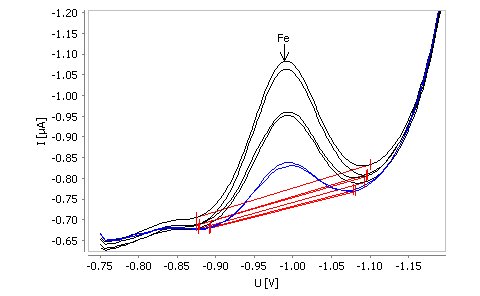
The voltammetric determination of the iron triethanolamine complex on the non-toxic Bi drop electrode does not require enrichment. This system uses catalytic signal enhancement, allowing both the detection at very low levels (limit of detection of 0.005 mg/L) and measurements in a wide range of concentrations up to 0.5 mg/L. This method is best suited for automated systems or process analyzers, allowing fully automatic determination of iron in a large sample series.
Sample
Drinking water, mineral water, sea water
Experimental
The water sample and the supporting electrolyte are pipetted into the measuring vessel. The determination of iron is carried out with a 884 Professional VA using the parameters specified in Table 1. The concentration is determined by two additions of an iron standard addition solution.
The Bi drop electrode is electrochemically activated prior to the first determination.

| Parameter | Setting |
|---|---|
| Mode | DP – Differential Pulse |
| Start potential | -0.75 V |
| End potential | -1.25 V |
| Peak potential Fe | -1 V |
Electrodes
- Working electrode: Bi drop
- Reference electrode: Ag/AgCl/KCl (3 mol/L)
- Auxiliary electrode: Glassy carbon rod
Results
The method is suitable for the determination of iron in water samples in concentrations from β(Fe) = 10–500 µg/L. The limit of detection of the method is approximately β(Fe) = 5 µg/L.
| Sample | Fe (µg/L) |
|---|---|
| Tap water spiked with β(Fe) = 20 µg/L | 19.1 |
References
Application Bulletin 439: Voltammetric determination of iron in water samples with a Bi drop electrode


 Share via email
Share via email
 Download PDF
Download PDF


