
AN-V-215
2020-05
Zinc in drinking water with the scTRACE Gold
Straightforward determination over a wide concentration range
Summary
At zinc concentrations above 3 mg/L, the quality of drinking water deteriorates, and an undesirable astringent taste is detectable. No health-based guideline value is required for zinc as it is an essential trace element for humans. The United States Environmental Protection Agency (US EPA) has set a maximum concentration of 5 mg/L as the limit value of Zn in drinking water.
The anodic stripping voltammetric method with an overall determination time of less than10 minutes is fast, very sensitive, and can be applied for a wide range of concentrations. Without applying any deposition time, the limit of detection is around 1 μg/L. This value can be lowered further when the deposition time is increased. When a reductive determination is carried out, the linear range of the method can be extended to 1.5 mg/L. The excellent performance of the method is due to the unique design and architecture of the scTRACE Gold electrode. This sensor does not require extensive maintenance such as mechanical polishing. Measurements can be performed in the laboratory or alternatively in the field. This method is suited for manual and automated systems.
Sample
Drinking water, mineral water
Experimental

The water sample and the supporting electrolyte are pipetted into the measuring vessel. The determination of zinc is carried out with the 884 Professional VA or with the 946 Portable VA Analyzer using the parameters specified in Table 1. The concentration is determined by two additions of a zinc standard addition solution.
The scTRACE Gold is electrochemically activated prior to the first determination.

| Parameter | Setting |
|---|---|
| Mode (884) Mode (946) |
DP – Differential Pulse SQW – Square wave |
| Start potential | -1.05 V |
| End potential | -0.45 V |
| Peak potential Zn | 0.06 V |
Electrodes
- scTRACE Gold
Results
Without deposition time, this method is suitable for the determination of zinc in water samples in concentrations from β(Zn) = 1–50 μg/L.
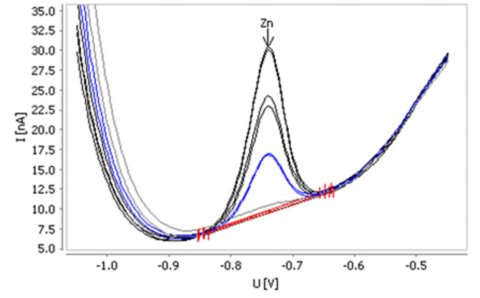
| Sample | Zn (μg/L) |
|---|---|
| Mineral water spiked with 10 μg/L Zn | 10.8 |
Internal references: AW VA CH4-0573-112018; AW VA CH4-0575-122018



 Share via email
Share via email
 Download PDF
Download PDF


